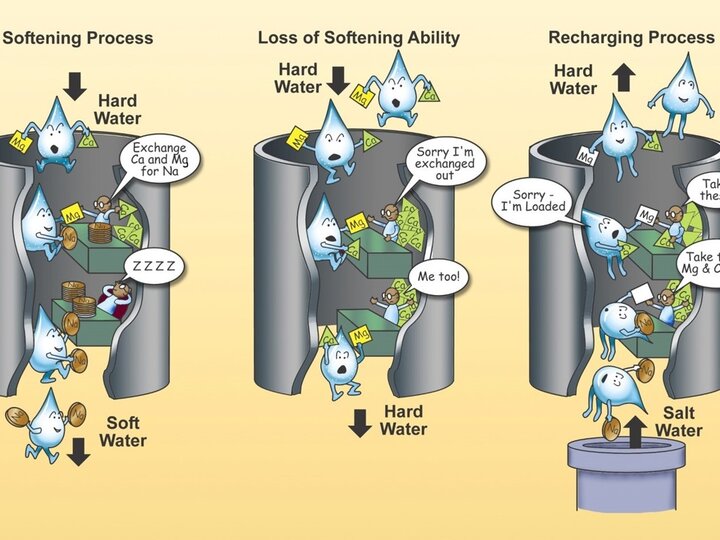
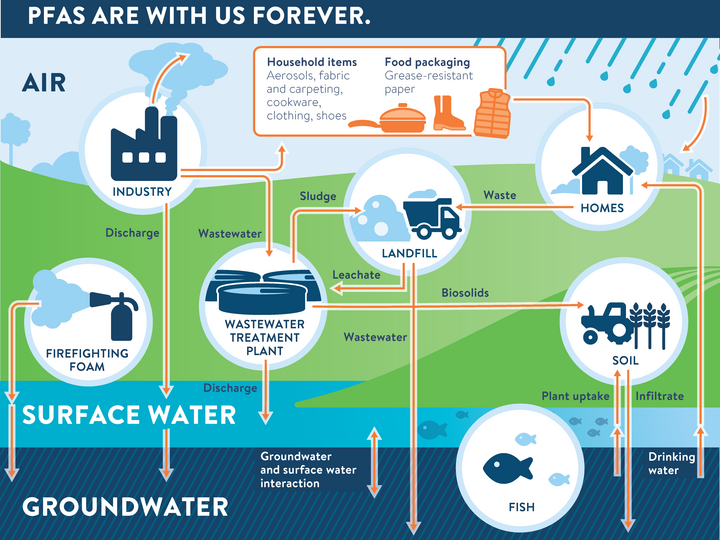
How much do you really know about the water you drink every day? Where does it come from? Is it safe to drink? Is a home water treatment system necessary? How can drinking water be protected? Explore this section for more detailed information that will help you answer questions or solve problems you may have.